
Laboratory Techniques and Measurements
Hands-On Labs, Inc. Version 42-0165-00-02
Lab Report Assistant
This report is not intended to replace an official laboratory report. The Lab Report Assistant is just a summarization of the questions, diagrams (where necessary), and data tables that are to be answered in a formal lab report. The goal is to enable students to write lab reports by simply providing this information in an editable file, which could be sent to an instructor.
Exercise 1: Length, Temperature, and Mass
Data Table 1. Length Measurements.
| Object | Length (cm) | Length (mm) | Length (m) |
| CD or DVD | 18 cm | 180 mm | 0.18 m |
| Key | 5 cm | 50 mm | 0.05 m |
| Spoon | 21 cm | 210 mm | 0.21 m |
| Fork | 15 cm | 150 mm | 0.15 m |
DataTable2. Temperature Measurements.
| Water | Temperature(°C) | Temperature(°F) | Temperature(K) |
| Hot from the tap | 34o C | 95o F | 308 K |
| Boiling | 97°C | 209°F | 367.15 K |
| Boiling for 5 minutes | 104°C | 218°F | 494 K |
| Cold from the tap | 16°C | 57°F | 287.15 K |
| Ice water – 1 minute | 11°C | 49°F | 282.15 |
| Ice water – 5 minutes | 5°C | 41°F | 278.15 K |
Data Table 3. Mass Measurements.
| Object | Estimated Mass (g) | Actual Mass (g) | Actual mass (kg) |
| Pen or pencil | 7g | 9g | 0.009 kg |
| 3 Pennies | 7.5 g | 7.5g | 0.0075 kg |
| 1 Quarter | 4 g | 5.7g | 0.0057 kg |
| 2 Quarters, 3 Dimes | 15 g | 18.2g | 0.0182 kg |
| 4 Dimes, 5 Pennies | 20 g | 22.1 g | 0.0221 kg |
| 3 Quarters, 1 Dime, 5 Pennies | 30 g | 32.6 | 0.0326 kg |
| Key | 6.5 g | 7.4 | 0.0074 kg |
| Key, 1 Quarter, 4 Pennies | 19.6 g | 23.1 | 0.0231 kg |
Questions
- At sea level, fresh water boils at 100 °C. Had the water in this experiment not boiled when it was put into a 100 °C environment, what might be the explanation?
**The boiling point of water is 100 °C in seawater. The water will boil at a lower temperature when at a level higher than the sea level because there is less pressure. When the water is lower than the sea level, it will boil at a higher temperature than 100 °C since it will be under increased air pressure.
- During the time of heating two different samples of water at sea level, one of them will boil at 102 o C, and the other will boil at 99.2 °C. Divide the difference between the theoretically calculated 100.0 o C and the experimental results (percent error) in each sample.
**The sample one with the boiling point of 102 degrees of celesials has the percent error of 2 percent. The percent error of sample two, which has a boiling point of 99.2 degrees Celsius, is -0.8 percent.
Exercise 2: Volume and Density
Data Table 4. Liquid Measurements.
| Mass A | Mass B | Mass B – A | ||||
| Liquid | Volume (mL) | Graduated Cylinder (g) | Graduated Cylinder withliquid (g) | Liquid (g) | Density g/mL | % Error |
| Water | 5.0 ml | 5.0 ml | 5.0 ml | 5.0 ml | 5.0 ml | 5.0 ml |
| Isopropyl alcohol | 19.4 g | 19.4 g | 19.4 g | 19.4 g | 19.4 g | 19.4 g |
Data Table 5. Magnet – Measurement Method.
| Object: | Mass (g) | Length (cm) | Width (cm) | Height (cm) | Volume (cm3) | Density (g/cm3) |
| Magnet | 4 g | 2.5 cm | 0.25 cm | 0.25 cm | 0.16 cm3 | 25 g/cm3 |
Data Table 6. Displacement Method.
| Object | Mass (g) | Initial volume of graduated cylinder (mL) | Final volume of graduated cylinder (mL) | Object Volume (mL) | Density (g/mL) |
| Magnet | 4 g | 8 ml | 10 ml | 2 ml | 2g/ml |
| Metal bolt | 7.6 g | 8 ml | 12 ml | 4 ml | 1.9g/ml |
Data Table 7. Archimedes’ method.
| Object | Mass (g) | Mass of Displaced Water (g) | Volume of Displaced Water (mL) | Density (g/mL) |
| Metal Bolt | 7.6 g | 117.5 g | 116.4 ml | 1.07g/ml |
| Magnet | 4 g | 117.1 g | 116.4 ml | 1.04 g/ml |
Questions
- The unknown, rectangular object is 3.6 cm in height, 4.21 cm in length, and 1.17 cm in width. Suppose that the mass is 21.3 g. What is the density of this substance (in grams per milliliter)?
** 1 gram = 1 cm 3.6 x 4.21 x 1.17 = 17.7 21.3g / 17.7mL = 1.2 g/mL The density is 1.2 g/mL
A sample of gold (Au) has a mass of 26.15 g. Since the theoretical density is 19.30 g/mL, what is the volume of the gold sample?
** Volume = Mass/Density Volume- 26.15 g /19.30 g/mL = 1.4 mL
- What would occur if you dropped the object into the beaker using the Archimedes Principle method instead of dropping the object into the beaker?
** In case you dropped an object in the beaker, but instead of submerging the object, you used the Archimedes Principle method. I think I would have the same result as the volume of the object, instead of a result that would be based on the buoyancy.
- How did the measurement of the density of the magnet by the Principle of Archimedes compare to measuring the density of the magnet by the calculated volume? What is the approach that could be more correct? Why?
** The measurements of the densities are nearly fifty percent less. The Archimedes Principle, the magnet, and the metal bolt seemed to have a lower density. The displacement method appeared to be more accurate because I did not hold the string to each submerged object. There is a great likelihood of human error.
- You are presented with a little piece of gold colored material and would like to know whether it is really gold. Applying the Archimedes Principle, you will find the volume of 0.40 cm 3 and the mass of 6.0 g. What inferences can you draw from your bare density analysis?
** To determine the authenticity, I would first find the actual density of gold (19.3 g/cm3 ) and then compute the density of the colored gold material and compare it to the density of the gold to verify authenticity. The numbers given for the gold colored object would make its density 15 g/cm3. I would then conclude that the gold colored object is not gold.
Exercise 3: Concentration, Solution, and Dilution
Data Table 8. Initial Concentration.
| Chemical | Mass ofVolumetric Flask | Mass of Sugar (g) | Molar Mass (g) | Moles in a Volumetric Flask | Total Volume (L) | Molarity (mol/L) |
| Sugar (C12H22O11) | 27.3 | 8 | 342.296 g | 0.079755 | 0.025 | 3.1902mol/L |
Data Table 9. Dilution Series.
| Dilution | Volume (mL) | Mass (g) | Density (g/mL) | Initial Concentration (M) | Volume Transferred (mL) | Final Concentration (M) |
| 0 | 25.0 mL | 27.7g | 1.108g/mL | 0 mL | 3.1902 M | |
| 1 | 25.0 mL | 24.9g | 0.996g/mL | 3.1902 M | 2.5 mL | 0.31902 M |
| 2 | 25.0 mL | 25.4g | 0.984g/mL | 3.1902 M | 4.5 mL | 1.741444 M |
| 3 | 25.0 mL | 25.1 | 1.004g/mL | 3.1902 M | 3.0 mL | 2.612166 M |
| 4 | 25.0 mL | 25.6 | 1.024g/mL | 3.1902 M | 6.0 mL | 1.306083 M |
Data Table 10. Molarity vs. Density.
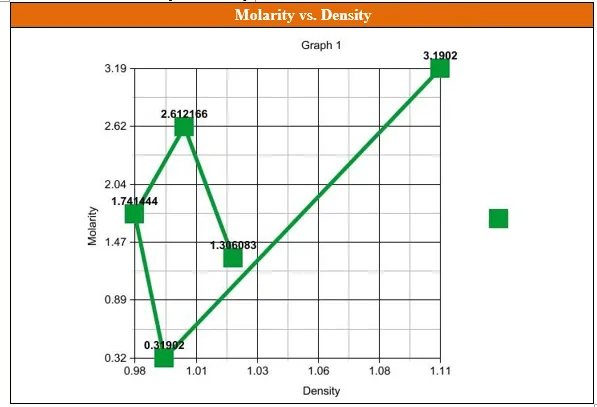
Questions
What would prepare 10 mL of a solution of HCl with a concentration of 0.25M, if 1M HCl is available? What is the required amount of 1M HCl? What is the amount of distilled water?
0.25M is 0.25 moles/L 0.25*10/1= 2.5 mL needed
- From the graph of Molarity vs. Density, created in Data Table 10, what was the relationship between the molarity of the sugar solution and the density of the sugar solution?
**The less the density of the sugar solution, the less the molarity. It was the same way round.
